Graphene supercapacitors
Graphene is a thin layer of pure carbon, tightly packed and bonded together in a hexagonal honeycomb lattice. It is widely regarded as a âwonder materialâ because it is endowed with an abundance of astonishing traits: it is the thinnest compound known to man at one atom thick, as well as the best known conductor. It also has amazing strength and light absorption traits and is even considered ecologically friendly and sustainable as carbon is widespread in nature and part of the human body.
Graphene is often suggested as a replacement for activated carbon in supercapacitors, in part due to its high relative surface area (which is even more substantial than that of activated carbon). The surface area is one of the limitations of capacitance and a higher surface area means a better electrostatic charge storage. In addition, graphene based supercapacitors will utilize its lightweight nature, elastic properties and mechanical strength.
A Graphene supercapacitor is said to store almost as much energy as alithium-ion battery, charge and discharge in seconds and maintain all this over tens of thousands of charging cycles. One of the ways to achieve this is by using a a highly porous form of graphene with a large internal surface area (made by packing graphene powder into a coin-shaped cell and then dry and press it).
What are supercapacitors?
Supercapacitors, also known as EDLC (electric double-layer capacitor) or Ultracapacitors, differ from regular capacitors in that they can store tremendous amounts of energy.
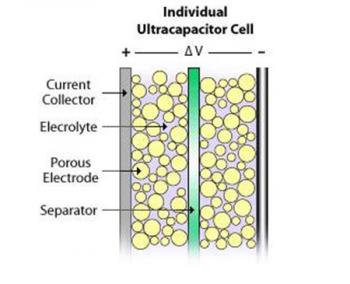
A basic capacitor usually consists of two metal plates, separated by an insulator (like air or a plastic film). During charging, electrons accumulate on one conductor and depart from the other. One side gains a negative charge while the other side builds a positive one. The insulator disturbs the natural pull of the negative charge towards the positive one, and that tension creates an electric field. Once electrons are given a path to the other side, discharge occurs.
Supercapacitors also contain two metal plates, only coated with a porous material known as activated carbon. They are immersed in an electrolyte made of positive and negative ions dissolved in a solvent. One plate is positive and the other is negative. During charging, ions from the electrolyte accumulate on the surface of each carbon-coated plate. Supercapacitors also store energy in an electric field that is formed between two oppositely charged particles, only they have the electrolyte in which an equal number of positive and negative ions is uniformly dispersed. Thus, during charging, each electrode ends up having two layers of charge coating (electric double-layer).
Batteries and Supercapacitors
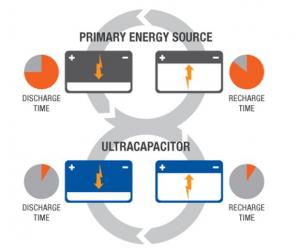
Unlike capacitors and supercapacitors, batteries store energy in a chemical reaction. This way, ions are inserted into the atomic structure of an electrode, instead of just clinging to it like in supercapacitors. This makes supercapacitors (and storing energy without chemical reactions in general) able to charge and discharge much faster than batteries. Due to the fact that a supercapacitor does not suffer the same wear and tear as a chemical reaction based battery, it can survive hundreds of thousands more charge and discharge cycles.
Supercapacitors boast a high energy storage capacity compared to regular capacitors, but they still lag behind batteries in that area. Supercapacitors are also usually more expensive per unit than batteries. Technically, it is possible to replace the battery of a cell phone with a supercapacitor, and it will charge much faster. Alas, it will not stay charged for long. Supercapacitors are very effective, however, at accepting or delivering a sudden surge of energy, which makes them a fitting partner for batteries. Primary energy sources such as internal combustion engines, fuel cells and batteries work well as a continuous source of low power, but cannot efficiently handle peak power demands or recapture energy because they discharge and recharge slowly. Supercapacitors deliver quick bursts of energy during peak power demands and then quickly store energy and capture excess power that's otherwise lost. In the example of an electric car, a supercapacitor can provide needed power for acceleration, while a battery provides range and recharges the supercapacitor between surges.
Common supercapacitor applications
Supercapacitors are currently used to harvest power from regenerative braking systems and release power to help hybrid buses accelerate, provide cranking power and voltage stabilization in start/stop systems, backup and peak power for automotive applications, assist in train acceleration, open aircraft doors in the event of power failures, help increase reliability and stability of the energy grid of blade pitch systems, capture energy and provide burst power to assist in lifting operations, provide energy to data centers between power failures and initiation of backup power systems, such as diesel generators or fuel cells and provide energy storage for firming the output of renewable installations and increasing grid stability.
Rivaling materials
Several materials exist that are researched and suggested to augment supercapacitors as much (or even more than) graphene. Among these materials are: hemp, that was used by Canadian researchers to develop hemp fibers that are at least as efficient as graphene ones in supercapacitor electrodes, Cigarette filters, which were used by Korean researchers to prepare a material for supercapacitor electrodes that exhibits a better rate capability and higher specific capacitance than conventional activated carbon and even higher than N-doped graphene or N-doped CNT electrodes.
Graphene supercapacitors commercialization
Graphene supercapacitors are already on the market, and several companies, including Skeleton Technology, the CRRC, ZapGoCharger, and Angstron Materials are developing such solutions. Read our Graphene Supercapacitors market report to learn more about this exciting market and how graphene will effect it.
Further reading
The latest graphene supercapacitor news:
Astra Energy announces agreement with Sustainable Energy Technologies for graphene/lithium-ion supercapacitors
Astra Energy has announced a strategic partnership agreement with Sustainable Energy Technologies (”SETI”) to supply Astra with the SETI Power Pack (SPP), the Company’s next generation energy storage solution that is a hybrid Graphene/Lithium-ion Supercapacitor intended to replace the need for traditional batteries.
In the PR, it was stated that: "When coupled with SETI Power Packs, the Holcomb Energy Systems (“HES”) In-Line Power Generator (“ILPG”) and the HES Self-Sustaining Power Plant in particular, are optimized to provide the end user with near continuous energy generation capability. Installing SPPs in series with an ILPG will enhance the overall effectiveness across the charge and discharge cycles, resulting in maximized operating efficiency of the HES Self-Sustaining Power Plant".
Graphene-Info publishes a new edition of its Graphene Supercapacitors Market Report
Today we published a new edition of our Graphene Supercapacitors Market Report, with all the latest information. The supercapacitor market and industry is facing high demand and graphene is a pivotal material for this application. This new update includes new company profiles and updates from various projects.
Reading this report, you'll learn all about:
- The advantages of using graphene in supercapacitors
- Various types of graphene materials
- Market insights and forecasts
- What's on the market today
The report package also provides:
- A list of all graphene companies involved with supercapacitors
- Prominent research activity in this field
- Free updates for a year
This Graphene Supercapacitors market report provides a great introduction to graphene materials used in the supercapacitor market, and covers everything you need to know about graphene in this niche. This is a great guide for anyone involved with the supercapacitor market, nanomaterials, electric vehicles and mobile devices.
Skeleton & Martinrea to collaborate on batteries for refuse trucks
It was recently reported that Skeleton Technologies and the Canadian automotive supplier Martinrea International are cooperating to equip refuse collection vehicles for use in Paris and New York with Skeleton’s ‘SuperBattery’ technology.
Martinrea International develops and produces electric vehicle technologies for heavy-duty commercial vehicles through its subsidiary Effenco. The aim of the agreement with the Estonia-based Skeleton is to electrify fleets of refuse collection vehicles in Europe and North America, starting in New York City and Paris, and later to make the solution developed by both companies available worldwide. In autumn 2022, Shell had already become known as the first major customer for the graphene battery solution. Shell wants to use it to create electrification solutions for mining sites.
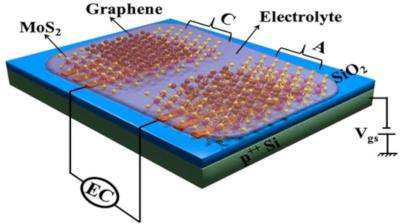
Researchers develop graphene/MoS2 micro-electrochemical capacitors for ultra-high charge storage
Researchers from the Indian Institute of Science have developed ultramicro-electrochemical capacitors with two-dimensional (2D) molybdenum disulphide (MoS2) and graphene-based electrodes. The development has great potential for wearables and implantable electronics as well as for sensors and miniature “smart” technology.
The miniature energy storage device uses graphene Flakes and MoS2 alternately in each electrode - the cathode and anode. Gel was used as the electrolyte, which makes it possible to integrate micro-supercapacitors into chips. This would be difficult if not impossible with a water-soluble electrolyte. The capacitor showed a capacitance of 1.8 mF/cm2 for a single-layer structure (graphene-MoS2). The multilayer electrode structure, consisting of multiple alternating layers of graphene and molybdenum disulfide, gained 30 times greater capacitance, or 54 μF/cm2.
The Graphene Flagship's chief, Prof. Jari Kinaret, shares his views on the latest graphene development and the post-Flagship future
The Enlit Europe 2022 energy conference recently took place, and the Graphene Flagship participated, showing some of the latest energy-related graphene projects. We took the chance to discuss graphene with some of the flagship researchers, and we also talked to Prof. Jari Kinaret, the director of the flagship project, to learn of how he summarizes the last 10 years now that the flagship project will soon end.
Q: We understand that the Graphene Flagship is attending Enlit Europe 2022, showing some new graphene R&D projects. We'll be happy to get an overview of what will be displayed at the event.
At Enlit Europe, the Graphene Flagship exhibited innovations from its Spearhead Projects, which are industry-led initiatives working to move materials from research labs towards commercial applications. Among these initiatives are:
- CircuitBreakers, led by ABB and developing first-of-their-kind grease-free, maintenance-free, low-voltage circuit breakers for fault protection in key parts of the electrical grid;
- GRAPES, led by Enel Green Power and working on combining silicon solar cells with perovskite solar cells, paving the way for low-cost, highly efficient photovoltaic energy, surpassing the limits of silicon based cells.
China startup Caiqi New Materials announced plans to mass-produce graphene supercapacitor material
Reports suggest that Chinese startup Caiqi Xin Cailiao (Caiqi New Materials) is preparing to mass-produce graphene materials for supercapacitors, in the near future.
According to these reports, Caiqi has already completed mass production trials and a fundraising round of about 10 million yuan ($1.4 million).
First Graphene and University of Manchester secure funding to advance graphene-enhanced supercapacitor material
First Graphene has announced it has secured grant funding, in conjunction with the University of Manchester (UoM), for the next stage of research into commercializing graphene-enhanced supercapacitor materials.
Awarded through Innovate UK’s “Accelerated Knowledge Transfer to Innovate” scheme (AKT2I),
the grant will be used to fund a project intended to accelerate development and optimization of a
graphene-metal-oxide slurry for manufacturing high energy density supercapacitors.
We released a new edition of our Graphene Supercapacitors Market Report
Today we published a new edition of our Graphene Supercapacitors Market Report, with all the latest information. The supercapacitor market and industry is facing high demand and graphene is a pivotal material for this application. This new updates includes a revamp of the general report, and updates from over 10 companies and research activities.
Reading this report, you'll learn all about:
- The advantages of using graphene in supercapacitors
- Various types of graphene materials
- Market insights and forecasts
- What's on the market today
The report package also provides:
- A list of all graphene companies involved with supercapacitors
- Prominent research activity in this field
- Free updates for a year
This Graphene Supercapacitors market report provides a great introduction to graphene materials used in the supercapacitor market, and covers everything you need to know about graphene in this niche. This is a great guide for anyone involved with the supercapacitor market, nanomaterials, electric vehicles and mobile devices.
Trans2Dchem project to develop improved graphene-based supercapacitors
TRANS2DCHEM, or “Transition of 2D chemistry-based supercapacitor electrode material from proof of concept to applications”, is a project that received € 2,485,717 from the EIC to create supercapacitors with increased energy density (above 50 Wh/L, which is about twice as much as the best components on the current market). This will allow their widespread use in electric cars as well as a battery support in devices that need to be supplied with large amounts of energy in a very short time.
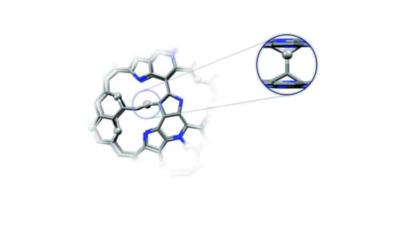
Theoretical model of GN3 structural fragment; image credit: CATRIN RCPTM
The researchers involved in the project utilize highly nitrogen doped graphene material (SC-GN3), offering energy density up to 200 Wh/L at a power of 2.6 kW/L, 170 Wh/L at 5.2 kW/L, and 143 Wh/L at 52 kW/L. The international team is developing supercapacitor prototypes, with manufacturing techniques that adhere to industry standards of leading supercapacitors manufacturers.
Researchers develop method for the fabrication of mesoporous graphene for energy storage systems
Researchers from South-Korea's Pohang University of Science and Technology (POSTECH), Kumoh National Institute of Technology, Sungkyunkwan University, Yeungnam University, Konkuk University and University of Seoul have proposed a simple strategy for the fabrication of mesoporous graphene with applications in high-performance energy storage systems like electric double-layer supercapacitors (EDLCs).
Conventional energy storage systems made of activated carbons (ACs) tend to have a poor power density due to the insufficient specific contact area, leading to inadequate creation of an electric double layer between electrode material and electrolyte. Therefore, an active material with a high specific contact area could help obtain high energy densities and meet the needs of various energy storage systems. Graphene's remarkable electrical conductance naturally makes it a logical candidate, but the high van der Waals contact between the graphene sheets makes stacking unavoidable, producing a limited available surface area.
Pagination
- Previous page
- Page 2
- Next page