Researchers from the US DOE's Pacific Northwest National Laboratory (PNNL) and Princeton University found a way to combine Graphene and indium tin oxide (ITO) nanoparticles to create cheaper and more durable fuel cells.
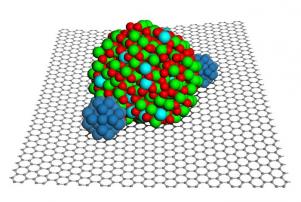
Fuel cells work by chemically breaking down oxygen and hydrogen gases to create an electrical current, producing water and heat in the process. The centerpiece of the fuel cell is the chemical catalyst — usually a metal such as platinum — sitting on a support that is often made of black carbon. A good supporting material spreads the platinum evenly over its surface to maximize the surface area with which it can attack gas molecules and is also electrically conductive.
The problem with using black carbon as support is that the platinum atoms tend to clump on such carbon, and water can degrade the carbon away. A different design uses metal oxides which are more stable and have better catalyst dispersion - but they have low conductivity and aren't easy to synthesize. The new design uses the best of both worlds...
The team is now incorporating the platinum-ITO-graphene material into experimental fuel cells to determine how well it works under real world conditions and how long it lasts.