A team of international researchers has made a graphene-based device that captures the real-time dynamics of a classic chemical reaction at the single molecule level. Developed at Peking University, UCLA and the Institute Chinese Academy of Sciences, the method could shed light on the mechanism of chemical and biological processes.
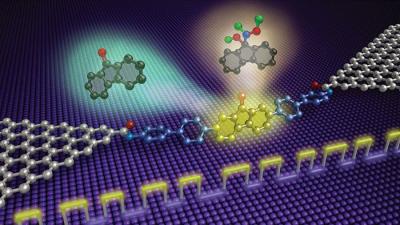
The device consists of two graphene arrays that flank a single molecule covalently tied to each array through amide linkers. The molecule, 9-fluorenone, contains a carbonyl group situated astride three fused rings. The team submerged the device in a solution containing a catalyst and the reagent hydroxylamine, which reacts with 9-fluorenone’s carbonyl group. The reaction changes the electrical charge of 9-fluorenone, so the team could follow the nucleophilic addition reaction by monitoring current conducted by the graphene arrays.
Unlike existing single-molecule methods, such as fluoresence microscopy-based ones, this new technique doesn’t rely on adding complex fluorescent labels to the molecule being studied. Also a key advantage of the graphene device is that researchers can monitor reactions with microsecond time resolution, says a researcher at Cornell University, who was not involved in the work. Microsecond time resolution is possible but challenging, for optical methods, he adds.