Researchers at Northwestern University have unveiled an intriguing new 2D material called borophene. It is a sheet of boron with a graphene-like honeycomb arrangement, but with an extra boron atom placed on top of each tessellated hexagon. Borophene has not yet been isolated as a free-standing sheet, but there are already indications that some of borophene’s properties could match, if not surpass, those of graphene and other 2D materials.
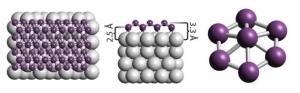
The researchers grew borophene on silver, by evaporating boron atoms from a solid boron rod at temperatures of 450ºC to 700ºC inside an ultrahigh-vacuum chamber. While boron itself is a poor electrical conductor, the scientists found that borophene is actually fully metallic - which is extraordinary amongst other 2D materials (that are usually semiconductors). Unprotected samples oxidized in a few hours, but a silicon covering kept them stable for several weeks.
Testing under a scanning tunnelling microscope revealed that borophene could take different forms, depending on the temperature used to make it and on how the atoms of boron sat on the silver below. One looked smooth, while the other appeared striped. Calculations suggest that corrugated borophene conducts electricity much better along these ridges than across them; it could even be stiffer and more conductive in this direction than is graphene. The researchers are now trying to measure these qualities experimentally, and say that their directionality could give borophene an advantage in certain applications, such as a switchable filter for polarized light.
The next goal is to demonstrate a free-standing sheet of borophene, which would allow researchers to accurately measure properties like conductivity. It seems, however, that this is far from being easy, as boron is highly reactive to substances around it. This tendency to react may eventually turn out to be an advantage, because borophene could be readily modified with other chemical groups or sandwiched between other materials to fine-tune its properties. Boron and many of its compounds are also extremely hard, so it might be easier to handle than more fragile 2D materials like silicene or germanene.
The researchers suggest that borophene has a higher electron density than graphene, which raises the possibility that cooled borophene could act as a superconductor, carrying electrical charge with no resistance.