Researchers from Stanford discovered that single-layer graphene is about 100 times more chemically reactive than double or triple sheets of graphene. The team bombarded single and multiple sheets of graphene (on silicon oxide substrates) with highly reactive hydrogen atoms generated in a stream of charged gas, or plasma. When they looked at the graphene afterwards, they found out that the single sheet of graphene was riddled with etch pits, but thicker layer were hardly pitted at all.
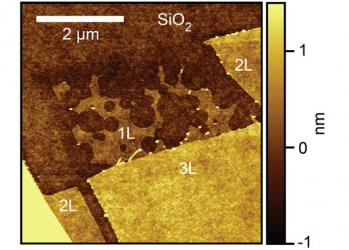
Those pits were caused by graphene's carbon atoms reacting with hydrogen atoms, presumably creating methane molecules that lift up and away out of the graphene sheet. The silicon oxide substrate is a participant in the etching reaction.
The researchers also noticed that the monolayer etch pits were circular, while the few etch pits found in thicker layers were hexagonal. They are not sure why, but it may be due to the substrate participation in the etch reaction. The next step in this research is to test the reactivity using different substrates, and maybe without a substrate at all.