Graphene Aerogel: Introduction and Market News
What is aerogel?
Aerogel is created by combining a polymer with a solvent to form a gel, and then removing the liquid from the gel and replacing it with gas (usually air). The high air content (99.98% air by volume) makes it one of the world's lightest solid material. Aerogels can be made from a variety of chemical compounds, and are a diverse class of materials with unique properties. They are known as excellent insulators, and usually have low density and low thermal conductivity.
Aerogels can be used in various applications, and although they have been around since the 1930s, their development is still progressing (for example, NASA's Glenn Research Center in Cleveland has invented several groundbreaking methods of creating new types of aerogels).
Common applications include enhancing the thermal performance of energy-saving materials and sustainable products for buildings, acting as a high performance additive to coatings, prevention of corrosion under insulation, uses in imaging devices, optics, and light guides, thermal breaks and condensation control, architectural lighting panels, outdoor and sports gear and clothing, and more.
Graphene aerogel
Graphene aerogel, also known as aerographene, is considered to be the least dense solid in existence (graphene aerogels are light enough to be balanced on small plants!).

Graphene aerogels are quite elastic and can easily retain their original form after some compression. In addition, the low density of graphene aerogels makes them very absorbent (to the point where it can even absorb more than 850 times its own weight). This means that it could be useful for environmental clean-ups like oil spills, and the aerogels only need to be picked up later after absorbing the spilled material. Graphene aerogel may also have some applications in both the storage and the transfer of energy by enabling the creation of lighter, higher-energy-density batteries - and vigorous research is being done on the matter.
Graphene aerogel are somewhat similar to graphene foams. Graphene foams are usually made by CVD growth on a metal structure (which is later removed), and are so more conductive than graphene aerogels.
Graphene aerogels are already being sold commercially, for about about $300 per gram.
Researchers develop fabrication strategy for improved graphene aerogels
While graphene aerogels have advantageous properties like extremely low weight, high porosity and good electrical conductivity, engineers who tried to use them to develop pressure sensors have encountered some difficulties.
Image credit: Nano Letters 2024
Specifically, many of these materials have an intrinsically stiff microstructure, which poses limits on their strain sensing capabilities. Researchers from Xi'an Jiaotong University, Northumbria University, UCLA, University of Alberta and other institutes recently introduced a new fabrication strategy for synthesizing aerogel metamaterials to overcome this limitation. This strategy fabricates a durable graphene oxide-based aerogel metamaterial that exhibits a remarkable sensitivity to human touch and motion.
Graphene Composites USA selected to join U.S Military footwear project
Graphene Composites USA (GC) has been selected to participate in a research and development program between DEVCOM Soldier Center, Natick MA and UMass Lowell, to develop materials for the next generation of U.S. military footwear.
The program, SWIFT [Supporting Warfighters through Innovative Footwear Technologies], is offered by the HEROES (Harnessing Emerging Research Opportunities to Empower Soldiers) initiative and will see GC extend its patented GC Composite graphene and aerogel technology to develop ultra-lightweight, durable, insulative materials for use in extreme cold weather.
Black Swan Graphene partners with Graphene Composites on graphene-enhanced ballistic protection technology
Black Swan Graphene has announced it has entered into a commercial partnership with Graphene Composites (GC). The Companies will aim to incorporate Black Swan's graphene in the fabrication of GC Shield, a patented ballistic protection technology ("GC Shields").
The Company highlighted that GC Shields, with its patented graphene-aerogel composite, have unique force dispersion capabilities which protect users from multiple shots, stacked rounds, and edge impacts while maintaining minimum back face deformation. They are among the strongest, lightest, and most resilient ballistic shields on the market for the law enforcement and defense sectors, according to Black Swan.
Researchers propose "Universal Murray's Law" for synthetic materials
Researchers from the University of Cambridge, Tokyo Institute of Technology, University of Warwick and University of Namur have proposed a new materials theory based on "Murray's Law," applicable to a wide range of hierarchical structures, shapes and generalized transfer processes.
The scientists experimentally demonstrated optimal flow of various fluids in hierarchically planar and tubular graphene aerogel structures to validate the proposed law. By adjusting the macroscopic pores in such aerogel-based gas sensors, they also showed a significantly improved sensor response dynamics.
Researchers develop formaldehyde sensing at room temperature graphene aerogels
Researchers at the University of Cambridge and the University of Warwick have developed a fully 3D-printed quantum dot/graphene-based aerogel sensor for highly sensitive and real-time recognition of formaldehyde at room temperature. Formaldehyde is a known human carcinogen that is a common indoor air pollutant. However, its real-time and selective recognition from interfering gases has thus far remained challenging, especially for low-power sensors suffering from noise and baseline drift.
The new sensor uses artificial intelligence techniques to detect formaldehyde in real time at concentrations as low as eight parts per billion, far beyond the sensitivity of most indoor air quality sensors.
Researchers develop graphene oxide-doped silica aerogels for efficient removal of pollutants from wastewater
Researchers from the Indian Institute of Technology Madras (IIT Madras) and Tel Aviv University in Israel, have developed a graphene oxide-doped silica aerogel adsorbent that can remove trace pollutants from wastewater.
This graphene-modified silica aerogel reportedly removes over 76% of trace pollutants (PPM level) in continuous flow conditions, offering a sustainable path for large-scale water purification. The research team is dedicated to enhancing these results for large-scale applications.
Stanford and UC Berkeley teams produce graphene aerogel in space
A team of researchers from Stanford University and the University of California, Berkeley are reportedly leveraging the International Space Station (ISS) National Laboratory to produce higher-quality graphene aerogel than is possible on Earth.
It was announced that this week, the Crew-6 astronauts onboard the space station completed work on the team’s investigation, which was funded by the U.S. National Science Foundation (NSF). The results could provide new insights into the underlying physics of graphene aerogel synthesis and lead to the development of novel material products.
Researchers create graphene hydrogels for efficient water purification
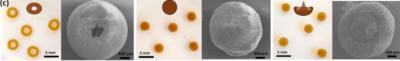
A team of researchers, led by Professor Aravind Vijayaraghavan based in the National Graphene Institute (NGI), have produced 3D particles made of graphene that come in various interesting shapes, using a variation of the vortex ring effect. These particles have also been shown to be exceptionally efficient in adsorbing contaminants from water, thereby purifying it.
Optical and SEM images of donut, spherical and jellyfish morphologies of GO-VR
The researchers have shown that the formation of these graphene particles is governed by a complex interplay between different forces such as viscosity, surface tension, inertia and electrostatics. Prof Vijayaraghavan said: “We have undertaken a systematic study to understand and explain the influence of various parameters and forces involved in the particle formation. Then, by tailoring this process, we have developed very efficient particles for adsorptive purification of contaminants from water”.
Graphene-enhanced composites and adhesives tested for use in satellites
An ESA project with Adamant Composites in Greece tested how the addition of graphene (and other nano-sized materials) can optimize a satellite’s thermal and electrical properties.
The airless vacuum of space is a place where a satellite can be hot and cold at the same time, with part of it in sunlight and the rest in the shade. Scientists work to minimize temperature extremes within a satellite’s body, because heat buildups might lead to parts going out of alignment or even buckling. Another undesirable outcome in highly-insulating vacuum conditions is for satellite surfaces to build up electrical charge, which may eventually result in disruptive or damaging discharge events. Composite materials are increasingly supplanting traditional metal parts aboard satellites, but these polymer-based materials possess lower thermal and electrical conductivity, compounding such problems.
Researchers develop a graphene oxide aerogel foam to improve all solid state lithium batteries
Researchers from China's Jiangsu University and Changzhou University have developed a non-ceramic solid-state electrolyte based on a graphene oxide (GO) aerogel framework filled with polyethylene oxide.
Li-ion batteries currently perform a key role in electric cars, but contain combustible liquid electrolytic materials which may pose major safety issues. The solid-state electrolyte (SSE) can replace the standard organic liquid electrolyte and is predicted to address safety issues such as leaking, heat runaway, and even explosions during operation.
Pagination
- Page 1
- Next page