Graphene and water treatment: introduction and market status
What is water treatment?
Water treatment is the collective name for a group of mainly industrial processes that make water more suitable for its application, which may be drinking, medical use, industrial use and more. A water treatment process is designed to remove or reduce existing water contaminants to the point where water reaches a level that is fit for use. Specific processes are tailored according to intended use - for example, treatment of greywater (from bath, dishwasher etc.) will require different measures than black water (from toilets) treatment.

Main types of water treatments
All water treatments involve the removal of solids (usually by filtration and sedimentation), bacteria, algae and inorganic compounds. Used water can be converted into environmentally acceptable water, or even drinking water through various treatments.
Water treatments roughly divide into industrial and domestic/municipal. Industrial water treatments include boiler water treatment (removal or chemical modification of substances that are damaging to boilers), cooling water treatment (minimization of damage to industrial cooling towers) and wastewater treatment (both from industrial use and sewage). Wastewater treatment is the process that removes most of the contaminants from wastewater or sewage, producing a liquid that can be disposed to the natural environment and a sludge (semi-solid waste).
Wastewater treatments usually consist of three levels: a primary (mechanical) level, in which about 50-60% of the solids are removed from raw sewage by screening and sedimentation, a secondary (biological) treatment level in which dissolved organic matter that escaped primary treatment is removed by microbes that consume it as food and convert it into carbon dioxide, water and energy. The tertiary treatment removes any impurities that are left, producing an effluent of almost drinking-water quality. Disinfection, typically with chlorine, can sometimes be an additional step before discharge of the effluent. It is not always done due to the high price of chlorine, as well as concern over health effects of chlorine residuals.
Seawater desalination are processes that extract salt from saline water, to produce fresh water suitable for drinking or irrigation. While this technology is in use and also holds much promise for growing in the future, it is still expensive, with reverse osmosis technology consuming a vast amount of energy (the desalination core process is based on reverse osmosis membrane technology).
What is graphene?
Graphene is a two dimensional mesh of carbon atoms arranged in the form of a honeycomb lattice. It has earned the title “miracle material†thanks to a startlingly large collection of incredible attributes - this thin, one atom thick substance (it is so thin in fact, that you'll need to stack around three million layers of it to make a 1mm thick sheet!) is the lightest, strongest, thinnest, best heat-and-electricity conducting material ever discovered, and the list does not end there. Graphene is the subject of relentless research and is thought to be able to revolutionize whole industries, as researchers work on many different kinds of graphene-based materials, each one with unique qualities and designation.

Graphene and water treatment
Water is an invaluable resource and the intelligent use and maintenance of water supplies is one of the most important and crucial challenges that stand before mankind. New technologies are constantly being sought to lower the cost and footprint of processes that make use of water resources, as potable water (as well as water for agriculture and industry) are always in desperate demand. Much research is focused on graphene for different water treatment uses, and nanotechnology also has great potential for elimination of bacteria and other contaminants.
Among graphene's host of remarkable traits, its hydrophobia is probably one of the traits most useful for water treatment. Graphene naturally repels water, but when narrow pores are made in it, rapid water permeation is allowed. This sparked ideas regarding the use of graphene for water filtration and desalination, especially once the technology for making these micro-pores has been achieved. Graphene sheets (perforated with miniature holes) are studied as a method of water filtration, because they are able to let water molecules pass but block the passage of contaminants and substances. Graphene's small weight and size can contribute to making a lightweight, energy-efficient and environmentally friendly generation of water filters and desalinators.
It has been discovered that thin membranes made from graphene oxide are impermeable to all gases and vapors, besides water, and further research revealed that an accurate mesh can be made to allow ultrafast separation of atomic species that are very similar in size - enabling super-efficient filtering. This opens the door to the possibility of using seawater as a drinking water resource, in a fast and relatively simple way.
Further reading
- Introduction to graphene
- Graphene company database
- How to invest in the graphene revolution
- The Graphene Handbook, our very own guide to the graphene market
Salgenx expands potential applications of its graphene and hard carbon-coated sand
Salgenx, developer of saltwater flow battery technology, has announced the expansion of its development of graphene and hard carbon-coated sand. Originally designed for use in advanced battery systems, Salgenx is now exploring a wide array of new applications for this innovative material, ranging from smart infrastructure to environmental sustainability.
The Company says that "the unique combination of graphene and hard carbon in a sand aggregate has the potential to transform the construction industry". By enhancing the electrical conductivity, mechanical strength, and durability of concrete, Salgenx’s carbon-coated sand opens up new possibilities in building and infrastructure development.
NematiQ GO membrane receives WaterMark Certification
Australia-based NematiQ has announced that after more than a decade of work, the NematiQ Graphene Oxide membrane has obtained WaterMark certification, solidifying its status as a safe product for water filtration.
The Australian WaterMark Certification Scheme is a mandatory scheme for plumbing and drainage products of a certain type. Certification ensures products are fit for purpose and appropriately authorized for use in plumbing and drainage installations. The Australian Building Codes Board administers and manages the Scheme.
ERDC and NASA to explore graphene-enhanced water treatment systems for space exploration
Researchers at the U.S. Army Engineer Research and Development Center (ERDC) are developing a water treatment system based on a mix of graphene oxide and a byproduct made from shrimp shells. Recently, ERDC's graphene research attracted the attention of NASA, leading to a collaboration with the agency to investigate the use of novel graphene materials as high-capacity absorbents for the removal of spacecraft water contaminants.
Graphene could significantly reduce the need for resupply and component sparing in space missions, potentially enabling crewed missions to deeper regions of space. Graphene-enhanced water treatment systems may one day be used to help extend future space missions and lead to safer intergalactic explorations.
Researchers develop bioinspired reinforced graphene membranes that overcome mechanical limitations
Researchers from Peking University, Beijing Normal University and KU Leuven recently reported a novel method to substantially reinforce large-area graphene membranes. Their work provides a facile method to fabricate large-area graphene membranes and paves the road to practical application in the membrane separation field.
Nanoporous graphene membranes are attractive for molecular separations, but it remains challenging to maintain sufficient mechanical strength during scalable fabrication and module development. In this work, the team drew inspiration from the composite structure of cell membranes and cell walls, and designed a large-area atomically thin nanoporous graphene membrane supported by a fiber-reinforced structure with strong interlamellar adhesion. It was found that factors like fracture stress, fracture strength, and tensile stiffness of the composite membranes can be enhanced compared with other graphene-based membranes of large scale.
Researchers develop graphene oxide-doped silica aerogels for efficient removal of pollutants from wastewater
Researchers from the Indian Institute of Technology Madras (IIT Madras) and Tel Aviv University in Israel, have developed a graphene oxide-doped silica aerogel adsorbent that can remove trace pollutants from wastewater.
This graphene-modified silica aerogel reportedly removes over 76% of trace pollutants (PPM level) in continuous flow conditions, offering a sustainable path for large-scale water purification. The research team is dedicated to enhancing these results for large-scale applications.
Evove completes pilot demonstration of graphene-enhanced filtration membranes at AB InBev brewery in South Africa
U.K.-based Evove, which develops graphene-enhanced membrane and filtering solutions, has announced it has completed a pilot demonstrating the use of its filter technology at a large AB InBev brewery in Johannesburg, South Africa.
It was explained that making beer is a process that requires a lot of water: Just sterilizing the equipment in a brewery can mean using thousands of gallons of water every week. That’s especially problematic in a place like drought-prone Johannesburg, where a water shortages and leaky infrastructure mean that water can be in short supply. Normally, the hot, caustic water used to clean tanks and water lines at a brewery can’t be reused, and ends up in the sewer system. It’s filled with grain residues, sugars, and yeasts that can’t easily be filtered out. “With conventional filters, it just clogs up the filters right away,” says Andrew Walker, Evove’s chief marketing officer.
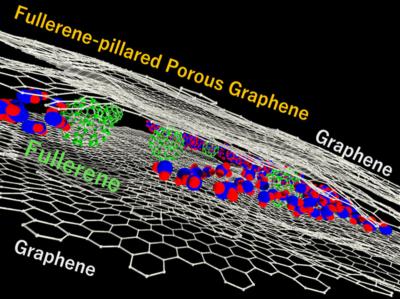
Researchers develop fullerene-pillared porous graphene material with high water adsorption capacity
A team of researchers from Japan's Chiba University, led by Associate Professor Tomonori Ohba and including master’s students, Mr. Kai Haraguchi and Mr. Sogo Iwakami, has fabricated fullerene-pillared porous graphene (FPPG)—a carbon composite comprising nanocarbons—using a bottom-up approach with highly designable and controllable pore structures.
Separation processes are essential in the purification and concentration of a target molecule during water purification, removal of pollutants, and heat pumping. To make the separation processes more energy efficient, improvement in the design of porous materials is necessary. Porous carbon materials offer a distinct advantage as they are composed of only one type of atom and have been well-used for separation processes. They have large pore volumes and surface areas, providing high performance in gas separation, water purification, and storage. However, pore structures generally have high heterogeneity with low designability, which poses various challenges, limiting the applicability of carbon materials in separation and storage.
Researchers develop rGO-based approach to address oil-water separation challenges
Researchers from Qatar University (QU), Maimoona Mohamed and Nada Yahya Deyab, along with their supervisor Dr. Shabi Abbas Zaidi, have made progress in addressing the challenge of oil-water separation.
Their research focuses on developing a novel material for efficient oil recovery from oil-water mixtures. By modifying polyurethane (PU) sponges and cotton with reduced graphene oxide (rGO), they have achieved promising results in terms of hydrophobicity, oil-absorption efficacy, reusability, and cost-effectiveness, offering a promising solution to address the issue of water and soil pollution caused by oil spills.
Researchers show that water can interact directly with electrons in graphene
An international team of researchers from the Max Plank Institute for Polymer Research of Mainz (Germany), the Catalan Institute of Nanoscience and Nanotechnology (ICN2, Spain) and the University of Manchester (England) has found that water can interact directly with the carbon’s electrons: a quantum phenomenon that is very unusual in fluid dynamics.
A liquid, such as water, is made up of small molecules that randomly move and constantly collide with each other. A solid, in contrast, is made of neatly arranged atoms that are surrounded by a cloud of electrons. The solid and the liquid worlds are assumed to interact only through collisions of the liquid molecules with the solid’s atoms: the liquid molecules do not “see” the solid’s electrons. Nevertheless, just over a year ago, a theoretical study proposed that at the water-carbon interface, the liquid’s molecules and the solid’s electrons push and pull on each other, slowing down the liquid flow: this new effect was called quantum friction. However, the theoretical proposal lacked experimental verification.
Directa Plus launches pilot project for treating produced water
Directa Plus has announced the start of a pilot project for a novel concept in produced water treatment. The Company said the project would use its ‘Grafysorber’ technology, and was being conducted through its subsidiary Setcar. The showcase of the pioneering endeavor reportedly took place at Setcar's Braila location in Romania
Produced water refers to the water that is extracted alongside crude oil during the oil production process. Currently, the water is disposed of without undergoing any treatment, but stricter industry regulations are driving the need for effective treatment solutions. Directa Plus said it was aiming to address the challenge with its new development, which had the potential to tap into untapped markets within the company's environmental division.
Pagination
- Page 1
- Next page