A team of researchers, led by Professor Sir Andre Geim at The University of Manchester, in collaboration with scientists from Belgium and China, used low-energy electrons to make individual atomic-scale holes in suspended graphene. The holes came in sizes down to about two angstroms, smaller than even the smallest atoms like helium and hydrogen.
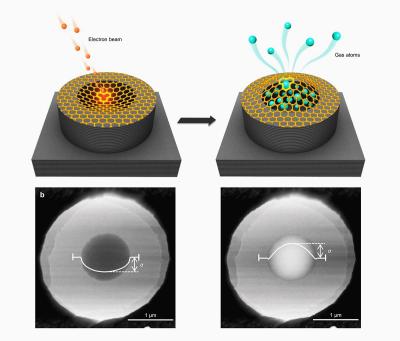
The researchers report that they achieved practically perfect selectivity (better than 99.9%) for such gases as helium or hydrogen with respect to nitrogen, methane or xenon. Also, air molecules (oxygen and nitrogen) pass through the pores easily relative to carbon dioxide, which is >95% captured.
Precision sieves for gases are certainly possible and, in fact, they are conceptually not dissimilar to those used to sieve sand and granular materials. However, to make this technology industrially relevant, we need membranes with densely spaced pores, not individual holes created in our study to prove the concept for the first time. Only then are the high flows required for industrial gas separation achievable, says Dr. Pengzhan Sun, a lead author of the paper.
The research team now plans to search for such two-dimensional materials with large intrinsic pores to find those most promising for future gas separation technologies. For example, there are various graphynes, which are also atomically thin allotropes of carbon but not yet manufactured at scale. These look like graphene but have larger carbon rings, similar in size to the individual defects created and studied by the Manchester researchers. The right size may make graphynes perfectly suited for gas separation.

