Researchers from Florida State University, Penn State University, Tsinghua University in China and the Institute of Carbon Science and Technology in Japan have come to fascinating conclusions on how to produce pure hydrogen, a green energy fuel by splitting water.
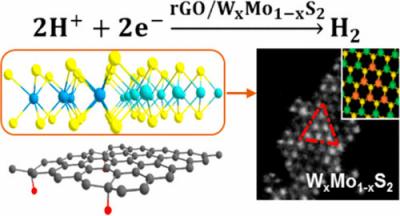
After experimenting with ways to use the compound molybdenum disulfide to split water, the team realized that the compound’s protons did not overlap well with that of hydrogen. They ultimately determined that the best way to split the hydrogen was to create an alloy with the molybdenum disulfide. They created a thin film with alternating graphene and tungsten-molybdenum layers.
According to the team, this created a much more efficient process and lowered the electrical voltage required to split the water from 200 millivolts with pure molybdenum disulfide to 96 millivolts. This has the promise of being as good as platinum, the researchers said.
Posted: Jul 10,2017 by Roni Peleg