Bionic mushroom interacts with bacteria and graphene to generate electricity
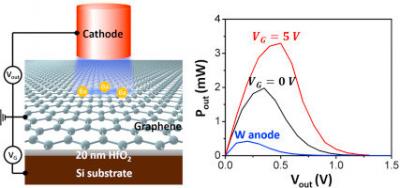
In a recent study, researchers from the Stevens Institute of Technology in the U.S have come up with an original idea - they designed a bionic mushroom that uses graphene to produce electricity. More accurately, the researchers have generated mushrooms patterned with energy-producing bacteria and an electrode network.

Many examples of organisms that live closely together and interact with each other exist in nature. In some cases, this symbiotic relationship is mutually beneficial. The research team wanted to engineer an artificial symbiosis between button mushrooms and cyanobacteria. In their vision, the mushroom would provide shelter, moisture and nutrients, while bacteria 3D-printed on the mushroom's cap would supply energy by photosynthesis. Graphene nanoribbons printed alongside the bacteria could capture electrons released by the microbes during photosynthesis, producing bio-electricity.